- Foundation
- Actions
- Osteoarthritis
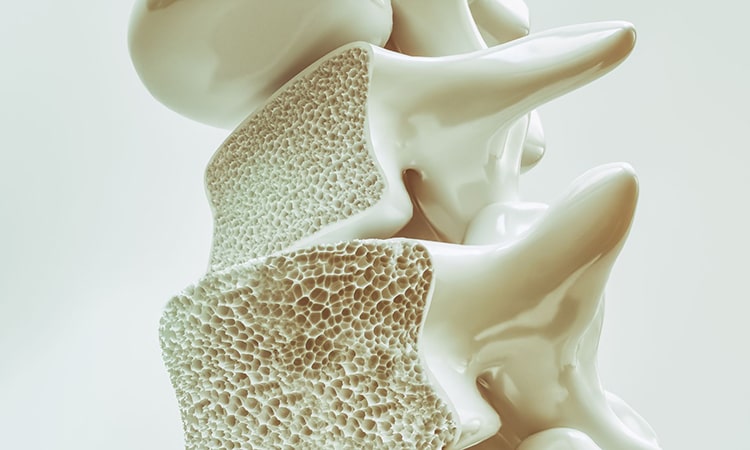
- Osteoporosis
- Actuality
- OAFI Radio/TV
- Get Involved
- Contact
-
-
-
OAFI
Osteoarthritis International FoundationC/ Tuset, 19 · 3º 2ª
08006 Barcelona
(+34) 931 594 015
info@oafifoundation.comSchedule:
Monday-Thursday 9AM-6PM
Friday 8AM-3PM
-
-
-

-

-

A study promoted by OAFI states that SYSADOAs are considered appropriate in the treatment of osteoarthritis

The journal of the Spanish Society of Rheumatology (SER), Clinical Rheumatology, has just published part of the results of a study promoted by the International Foundation for Patients with Osteoarthritis, OAFI, which clarifies the use of SYSADOAs in the treatment of Osteoarthritis in Primary Care: “Expert Paper on the Appropriate Use of SYSADOAs in Controversial Clinical Situations.”
Oral SYSADOA (SYMPTOMATIC slow-acting drugs for osteoarthritis) are natural compounds that have been shown to be useful and safe in the treatment of osteoarthritis.
However, the discrepant results observed in certain studies and meta-analyses and the limited or non-existent evidence on the use of oral SYSADOAs in certain patient profiles and clinical situations have generated controversy regarding its use. Not all patients respond to oral SYSADOAs and discrepancy in outcomes and lack of evidence in some clinical situations has resulted in different recommendations in the different therapeutic guidelines. Heterogeneity in the recommendations for the use of oral SYSADOAs and, consequently, in the prescription by specialists, may result in inappropriate use of these drugs.
OAFI decided to convene a scientific committee composed of 6 internationally recognized clinical experts and a group of 15 clinical experts in the world of osteoarthritis and recommended by the Primary and Specialist Medical Societies (SEMG, SEMERGEN, SETRADE, SemFYC , SER) to ask for their opinion based on clinical experience and scientific evidence on the appropriate use of SYSADOAs using the Delphi technique.
During two rounds of questions, 206 specific consultations structured in 24 clinical questions were assessed.
The results were analysed and discussed in a meeting attended by all experts and during which three important points were agreed.
(1) Individual patient factors such as weight, age, physical activity, etc. may condition the use of oral SYSADOAs;
(2) Oral SYSADOAs, especially chondroitin sulfate and its combination with glucosamine, are considered appropriate in primary osteoarthritis (knee, hand, and hip) and in some types of secondary osteoarthritis; they are not considered appropriate in erosive osteoarthritis of the hands, shoulder, spine, and ankle.
(3) Oral SYSADOAs may be prescribed to patients at risk or associated chronic disease being in most cases the only therapeutic alternative.
Through this work, it has been possible to know the position of a group of experts in the face of clinical situations for which there is no scientific evidence regarding their use.
We hope that this work can contribute to improving the protocols for the use of SYSADOAs in the treatment of osteoarthritis and be a useful tool in situations of uncertainty.
You can consult the paper published in the Journal of the Spanish Society of Rheumatology here. You can also find more information about the study here, with the detailed study and the OAFI Radio programs in which we talk about it.


























Leave a Reply